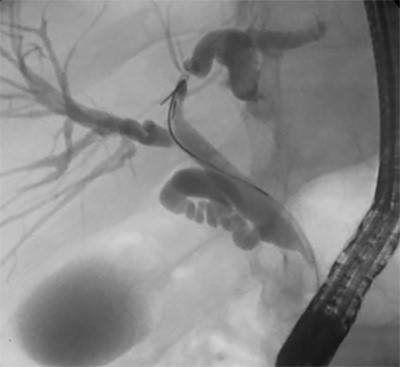
Once the EKOS catheters were adequately placed, the catheter and sheaths were secured and connected to tissue plasminogen activator infusion ports and insertion of the ultrasound intraluminal core. Intraoperative the intraluminal core is activated by a control unit to emit high frequency (2.2 MHz) and low-power (3.5 W) ultrasonic energy. A continuous tPA rate of 0.5mg/hr/catheter and normal saline coolant rate of 35ml/hr/catheter was started and continued postoperatively.
The patient tolerated the procedure well and transported to the ICU in stable condition. In the surgical ICU, the heparin drip was transitioned to 250units/hr and goal PTT <40. Fibrinogen levels every 6 hours were followed with the target fibrinogen >200 and if <150 the tPA rate would be decreased by half. The patient’s fibrinogen levels throughout the postoperative period range from 170-183 and did not require any adjustment of the tPA rate.
On POD 1, approximately 30 hours after EKOS catheters placement, the patient underwent a transthoracic echocardiogram that showed resolution of right heart strain, with RV/LV of 0.88 from 1.8. The patient was bedrest during the postoperative catheter infusions, however endorse resolution of the shortness of breath. The EKOS catheters were removed 1 hour after the echocardiogram. Pressure was held for 30 minutes at the puncture sites and did not require additional medical management. Approximately 39 hours since the time of procedure the patient was hemodynamically normal, afebrile, resolution of SOB, no oxygen requirement, ambulating without leg pain, and deemed appropriate for discharge. The patient was started on Rivaroxaban (Xarelto) and received initial follow-up the with anticoagulation clinic and primary care provider. The patient was seen in vascular surgery clinic one month later with an echocardiogram that continued to show normal RV function with return to baseline health.
Discussion
Acute pulmonary embolism (PE) is a common event resulting in 100,000–180,000 deaths per year in the United States.2,3 However, a subset of patients may present with right ventricular (RV) compromise or hemodynamic instability requiring acute intervention to decrease clot burden.
Systemic thrombolysis is the standard of treatment for massive PE, leading to improvement of RV function and hemodynamic status. However, is associated with a high risk of major bleeding complications (20 percent) and intracranial hemorrhage (3 percent).4 Therefore, alternative modalities have aimed to be effective in reversing RV dilation and dysfunction and reducing adverse clinical events. The Food and Drug Administration approved procedural intervention for PE include surgical embolectomy, aspiration thrombectomy, and CDT. Surgical embolectomy has fallen out of favor due to lack of collective surgical expertise and medical comorbidities precluding surgery. Aspiration thrombectomy using a Greenfield suction embolectomy removes centrally located embolus by applying negative pressure to the tip by manual suction. However, the major disadvantage is the large lumen catheter is difficult to manipulate because of its size, rigidity and no guidewire assistance.2-4
Catheter directed thrombolysis with ultrasound-assisted have demonstrated excellent clinical results.5,6 The general aim of catheter-directed therapy in the setting of compromised hemodynamics or cardiac function with acute PE is to debulk or redistribute the obstructive clot.7 In recent years, the EkoSonic Endovascular System (EKOS) has risen in practice as a catheter based system that uses high frequency and lower-energy ultrasound waves to aid in the delivery and uptake of thrombolytic agent within the clot.8 The EkoSonic ultrasound-assisted catheter directed thrombolysis has been utilized to treat acute massive and submassive PE at multiple institutions.5, 8-10
The SEATTLE II and the ULTIMA studies looked at the clinical applications of CDT for PE patients with RV dysfunction or hemodynamically unstable. The studies demonstrated the improvement on RV function, decrease intracranial hemorrhage, and similar mortality and morbidity compared to systemic thrombolysis.3, 6,7 Yet, additional evidence and studies are needed in the short and long-term on CDT before the intervention is deemed the standard of care compared to systemic thrombolysis. However, CDT for patients with massive and submassive PE does play a role in clinical application.7,11 Currently, multiple institutions have used CDT for patients with submassive and massive PE or contraindications for systemic thrombolysis.6, 8-10 For the past two years our institution’s vascular surgery department has implemented the use of CDT for massive and submassive PE (Figure 5) based upon other institutions protocols that have a long-established track record of CDT in clinic practice.3,4,8-10
Evaluating right heart strain secondary from a PE is determinate for morbidity and mortality for PE patients and plays a major role of the application of CDT (Figure 5). The degree of right heart dysfunction is quantified using the right ventricle to left ventricle (RV/LV) ratio; a RV/LV ratio <0.9 is normal, 0.9-1.2 mild dysfunction, 1.2-1.5 moderate dysfunction, and >1.5 severe right heart dysfunction. The RV/LV is measured by CTA chest or echocardiogram. For patients with RV/LV >1.5 or hemodynamic unstable secondary from an acute PE, emergent catheter-directed thrombolysis should be considered as a possible intervention. Postoperatively all patients at our institution underwent an echocardiogram to evaluate RV function and improvement prior to removal of EKOS catheters.
The patient presented was noted to have acute submassive pulmonary embolism leading to severe RV strain with a RV/LV ratio of 1.8. Our patient postoperatively on day 1 was noted to have resolution of symptoms and underwent an echocardiogram that demonstrated an RV/LV of 0.88 with no signs of RV dysfunction. The patient was noted to be a very healthy individual and was discharged from the ICU to home with anticoagulation therapy on postoperative day 1.
The average hospital length of stay for massive and submassive PE patients who underwent CDT in literature review is (3.3 – 8.4) days.5,6,8,10 However, it should be noted that the treatment for massive PE skewed the average length of stay compared to submassive PE. The average length of stay for patient’s undergoing EKOS placement at our institution is 3.7 days. The patient presented was classified as a submassive PE with hemodynamically normal vitals, which has a significantly better morbidity and mortality compared to a massive PE. Based upon our patient having minimal comorbidities, postoperative normal RV/LV ratio, and resolution of symptoms made him an ideal candidate to be discharged on POD 1. It should be noted to be cautious and selective of patients’ to be discharged in an expedite manner post-procedural - particularly with patients with massive PE, hemodynamically unstable on presentation, and symptomatic post-procedural.
CDT in a meta-analysis has a major bleeding complication rate of 2.4 percent compared to 20 percent for systemic TPA.10 Even though, significantly lower than systemic thrombolysis, CDT has does carry a mild risk of intraoperative and post-procedural bleeding. Potential pitfalls and concerns for rapid patient discharge are the serious and minor complications associated with CDT. Notable severe complications include death, cardiac ischemia, stroke, and bleeding complication requiring surgical intervention. Notable minor complications include puncture site hematomas, arteriovenous fistula formation, and bleeding that interrupts or stops CDT without requiring transfusion or resuscitative support. Our patient progressed without any serious or minimal adverse events and therefore was a candidate to be discharged swiftly.