Abstract
Background
A 78-year-old male with a remote history of peptic ulcer disease presented with a history of ongoing abdominal pain, new-onset black, tarry stools, and radiological evidence of pneumatosis intestinalis with pneumoperitoneum.
Summary
In this case, a 78-year-old male patient on chronic low-dose steroids with a remote history of peptic ulcer disease presented with a history of abdominal pain refractory to initial medical management by a primary care physician. The presence of black, tarry stools prompted his presentation to the ED. Physical exam and labs did not indicate the need for acute intervention. However, radiological evidence of pneumatosis intestinalis with pneumoperitoneum was concerning for hollow viscus injury and potential bowel compromise. The patient underwent a non-therapeutic exploratory laparotomy with methylene blue chromoendoscopy, which did not reveal mucosal compromise or bowel injury. Pneumoperitoneum was attributed to rupture of emphysematous blebs along, but not within, bowel wall. He had an uncomplicated postoperative course, and extensive follow up with his primary care physician and gastroenterologist helped to determine medical interventions to mitigate persistent abdominal discomfort.
Conclusion
Pneumatosis intestinalis is a rare finding, and its significance cannot be determined outside of a clinical context. Methylene blue chromoendoscopy is a reliable adjunct technique for assessing mucosal compromise in cases of PI with pneumoperitoneum.
Key Words
pneumatosis; pneumatosis intestinalis; pneumoperitoneum; exploratory laparotomy
Case Description
Pneumatosis intestinalis (PI) is a rare finding that may accompany benign, self-limiting processes or signal potentially fatal circumstances that require urgent surgical intervention. Because it is a pathological sign and not a diagnosis itself, PI may be associated with various underlying etiologies, from benign, self-limiting conditions to fatal ones. Moreover, PI can be caused by these pathologies, as well as a benign idiopathic condition1,2 or iatrogenic causes.3,4 There are multiple theories proposed for the mechanism by which patients develop PI, which is not mutually exclusive. They include bacterial overgrowth causing increased intraluminal gas, increases in intraluminal gas secondary to blunt trauma, mucosal compromise due to corticosteroid-induced atrophy of Peyer’s patches, and pulmonary gas migration via vascular channels.5–7
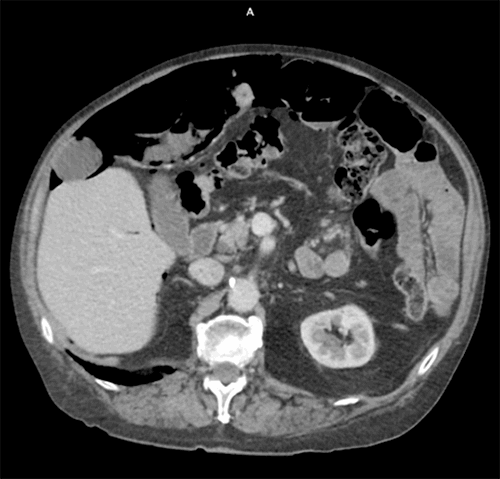
The case we present here describes a patient who warranted surgical intervention due to concomitant pneumoperitoneum. This case illustrates an example of radiologically identified PI with surgical intervention. A 73-year-old man presented to the emergency department (ED) with ten days of epigastric pain and one dark, tarry bowel movement the previous night prompting his ED visit. He had complained of epigastric pain to his primary care physician (PCP) just days prior, who started him on a proton pump inhibitor which failed to relieve his symptoms. He denied any other changes in bowel movements, and his medical history was significant for GI bleeding 25 years previous secondary to excessive NSAID use, which was diagnosed as peptic ulcer disease and quickly resolved after discontinuation. His most recent colonoscopy was one year before presentation with one benign polyp. He was adherent to a chronic course of prednisone 10 mg daily for the management of polymyalgia rheumatica.
On arrival, he was afebrile and hemodynamically stable. Physical exam revealed mild epigastric tenderness but no rebound, guarding, distension, masses, or organomegaly. On rectal exam, he had dark stool that was guiac negative. His labs were significant for white blood cell count of 9,600/μL, creatinine of 1.3 mg/dL, and lactic acid of 1.47 mmol/L. A CT scan was obtained, which showed pneumatosis of the small bowel and extensive pneumoperitoneum, and he was urgently taken to the OR for concern of hollow viscous injury with possible bowel ischemia (Figure 1).